Phoenix® WinNonlin® is the industry standard for non-compartmental analysis (NCA), pharmacokinetic/pharmacodynamic (PK/PD), and toxicokinetic (TK) modeling. Integrated tools for data processing, post-analysis processing, table creation, graphics, and compliance create an all-in-one collaboration workbench used by scientists, reviewers, medical writers, and quality assurance staff for drug development projects.
Jan 10, 2012 - Since a renewal fee of Phoenix WinNonlin increases tremendously every year, i would like to change program to analyze pharmacokinetic.
Relied on by Scientists and Regulators around the Globe
With a proven 30-year history, Phoenix WinNonlin is used by over 6,000 scientists at more than 1,500 establishments in 60 countries, including top pharmaceutical companies and academic institutes. Regulatory agencies, including 11 divisions of the US Food and Drug Administration (FDA), Japan Pharmaceutical and Medical Device Agency (PMDA), China Food and Drug Administration (CFDA), and the UK Medicines and Healthcare Products Regulatory Agency (MHRA), all use Phoenix WinNonlin to evaluate drug submissions.
Intuitive. Flexible. Sophisticated.
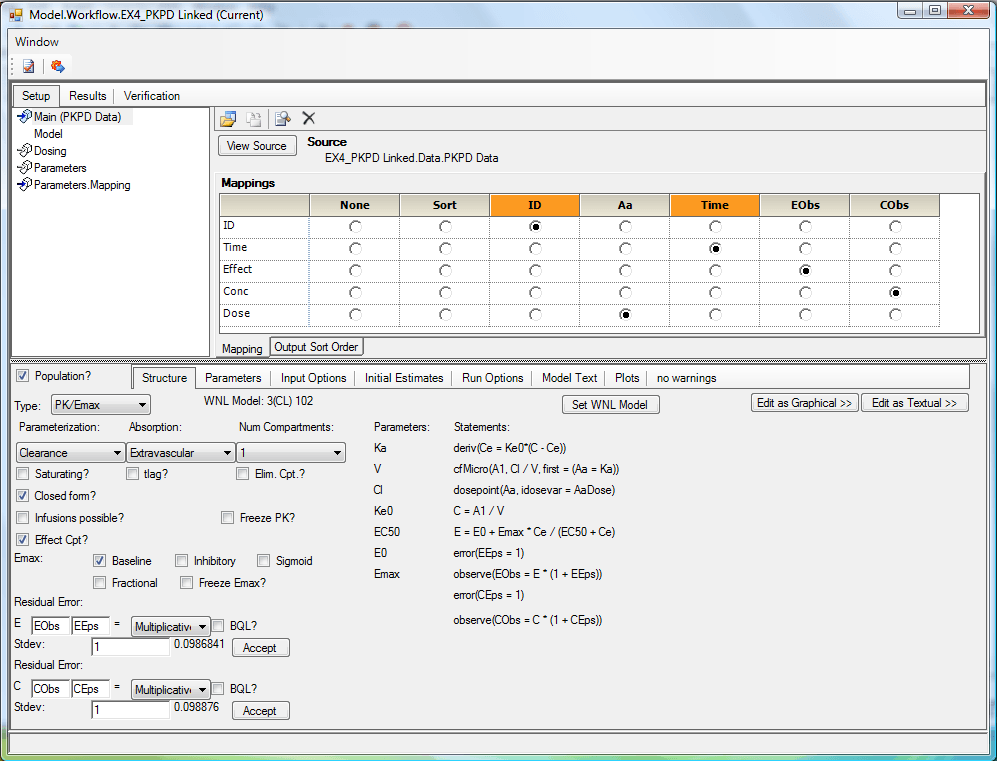
From beginners to advanced modelers, Phoenix WinNonlin provides the most comprehensive set of analysis tools for early non-clinical research to large clinical trial PK/PD studies.
- Our NCA and individual PK/PD modeling engine and statistical analysis tools can be used for a wide-range of studies and analyses
- The powerful NCA engine automatically outputs additional NCA parameters for plasma and urine to save time, reduce errors, and provide higher transparency for regulatory agencies
- Organizations can set strict criteria with user-defined parameters for calculating the terminal slope in NCA
- NCA ratios are automatically calculated increasing efficiency and saving time
Powerful Integrated Graphics Engine and Table Generator
The integrated graphics engine in Phoenix WinNonlin automatically creates publication-quality plots, figures and tables to create standardized PK/PD reports. Phoenix 8.1’s enhanced charting capabilities provide the ability to utilize the offset function, change font size and style, change the order and presentation of categorical axes, and more. The outputs can be quickly attached to a PK/PD report for communicating results internally and to regulatory agencies.
X-Categorical Plot with Order Change
Data Processing and Preparation Tools
Dataset preparation is intuitive with Phoenix WinNonlin’s data processing tools:
- Users can utilize the simple graphical user interface to import SDTM-formatted datasets and prepare analysis-ready datasets. The Phoenix workflow traces data from the moment it is imported through final analysis, preventing data loss and costly re-analysis.
- The new one-step ratios plugin provides quick and easy calculation of NCA ratios including bioavailability, accumulation, linearity index, and parent/metabolite comparisons.
- Our Enhanced Descriptive Statistics provide automated calculations for sample and population statistics for skewness and kurtosis, user-specified percentiles, and more.
Phoenix Tools to Support Compliance
Phoenix offers features and applications to ensure that organizations are compliant with organizational and regulatory policies:
- The combination of Phoenix WinNonlin with the Phoenix Knowledgebase Server (PKS) enables management of clinical and non-clinical PK/PD data and analyses in compliance with the US FDA electronic records and signatures regulation (21 CFR Part 11)
- Passwords can be used to lock workflows, ensuring consistency and quality control
- The integrated and enhanced Validation Suite for WinNonlin completes validation in minutes, not days. Robust reports and updated validation documents in Phoenix ensure alignment with regulatory guidance.
- Phoenix’s CDISC Workflow Templates automate the creation of SDTM or SEND Pharmacokinetic Parameters (PP) and Pharmacokinetic Concentration Data (PC) domains in the FDA-required SDTM or SEND format
We’re committed to your success
We provide a wide range of education and support tools to get you started, and our informative blog and instructive webinars keep you up to date with the most important advancements in modeling. The new Certara Professional Certification program offers online accreditation of user competency and proficiency analysis of PK/PD data using Phoenix WinNonlin 8.0 and 8.1.
Contact us to learn why Phoenix WinNonlin is the trusted, proven industry standard software tool for PK/PD modeling and non-compartmental and compartmental analysis.
ST. LOUIS--(BUSINESS WIRE)--Pharsight, a market-leading provider of software and scientific services to improve productivity and decision-making in clinical drug development, today announced the launch of Phoenix WinNonlin, the next generation of the company’s industry-standard software tool for pharmacokinetic and pharmacodynamic (PK/PD) modeling and noncompartmental analysis. Phoenix WinNonlin 6.0 is the cornerstone of Phoenix™, Pharsight's new desktop software platform that is designed to advance model-based drug development by providing an integrated environment for data analysis, modeling and simulation.
The major features of Phoenix WinNonlin 6.0 include a new graphical user interface that makes creation and re-use of PK/PD analysis workflows seamless and intuitive, powerful native graphics and improved data management tools. Phoenix WinNonlin will be backward-compatible with WinNonlin versions 4 and 5. Phoenix WinNonlin will continue to operate with Pharsight’s enterprise PK/PD data management system, Pharsight Knowledgebase Server™ (PKS™), and can also be used independently of PKS.
In addition to the release of Phoenix WinNonlin 6.0, newly released Phoenix Connect™ software integrates applications built on the Phoenix platform with commonly used third-party analysis and modeling tools such as S-PLUS®, NONMEM® and SAS®, and enables data import/export with emerging industry standards such as CDISC.
“The release of Phoenix WinNonlin represents an important milestone in Pharsight’s vision for software to enable model-based drug development and its utility in translational science,” said Daniel Weiner, Ph.D., senior vice president and chief technology officer of Pharsight. “Phoenix WinNonlin version 6.0 merges the trusted algorithms from WinNonlin with new powerful data management tools and high-quality graphics to support comprehensive PK/PD analysis. The Phoenix platform has been developed to provide a collaborative environment for drug development scientists and to shorten the learning curve to more complex and impactful modeling, which ultimately supports more confident and efficient clinical development decisions.”
Concurrent with the release of Phoenix WinNonlin 6.0, Pharsight has also released version 4.0 of Pharsight Knowledgebase Server™ (PKS™). PKS is an enterprise data management system for secure storage and tracking of PK/PD data and analyses that is tightly integrated with Phoenix WinNonlin and earlier WinNonlin versions, as well as Pharsight’s complementary software tools for PK analysis automation and reporting. The major features of PKS 4.0 include new web user interface enhancements for navigation of study and scenario data, improved data transfer utilities, and configurable data mining capabilities.
“Pharsight plans to release new Phoenix-enabled versions of all our products that process, analyze and report on pharmacokinetic and pharmacodynamic data to enable knowledge sharing and efficiencies across the PK/PD analytic spectrum,” added Dr. Weiner. “In particular, we have had an excellent early response to the beta program for Phoenix NLME™, our population PK/PD modeling tool currently under development that will optionally interface with Phoenix WinNonlin.”
About Phoenix™ WinNonlin®
Phoenix WinNonlin 6.0 is the next generation of Pharsight’s industry standard software for PK/PD modeling and noncompartmental analysis. Phoenix WinNonlin improves scientific productivity during critical drug development tasks by providing a comprehensive analysis environment that implements efficient reusable workflows and high quality outputs to reduce barriers to learning and collaboration and to enable more impactful quantitative decision making. More information about Phoenix WinNonlin is available at www.pharsight.com.
About Pharsight
Pharsight, a Certara™ company, is a market-leading provider of software products and scientific consulting services to help pharmaceutical and biotechnology companies improve their drug development process, regulatory compliance and strategic decision-making. Established in 1995, the company’s goal is to help customers reduce the time, cost and risk of drug development, as well as optimize the post-approval marketing and use of pharmaceutical products. Pharsight leverages expertise in its software tools and in the disciplines of pharmacology, drug and disease modeling, human genetics, biostatistics, strategic decision-making and regulatory strategy. Headquartered in St. Louis, Missouri, with more than 1200 customers worldwide, Pharsight products and services are used by all of the world’s top 50 pharmaceutical firms.
About Certara
Certara is dedicated to improving human health through a broad spectrum of products and services, from molecular discovery to clinical research. Formed in 2008, Certara unites Tripos and Pharsight Corporation. Tripos is a global leader in innovative scientific solutions enabling life science researchers to improve the efficiency of molecular discovery. Pharsight is the leading provider of software and scientific services to improve productivity and decision-making in clinical drug development. Certara expects to provide its clients with important new products, key enabling technologies and scientific expertise to expedite the flow of information throughout drug discovery and development. Certara is focused on reducing the barriers between the phases of research that will speed discoveries in chemistry and enable pharmaceutical and biotechnology companies to achieve significant and enduring improvements in the development and use of therapeutic products.
Registered Trademarks and Trademarks
Pharsight, Phoenix, Phoenix WinNonlin, Phoenix Connect, Phoenix Validation Suite, Phoenix NLME, WinNonlin, PKS and Pharsight Knowledgebase Server are trademarks or registered trademarks of Tripos, L.P. All other brands and product names are trademarks or registered trademarks of their respective holders.
